We firmly believe in the fact that the inhalation preparation is and will be the mainstream product in the market.
Our products will be approved in China and EU from year 2021 and on, and with a global sell, we will be a leading company in inhalation products and eyedrops soon.
Aureole's Salbutamol Sulfate Inhalation Solution Recieved CDE Reference Number
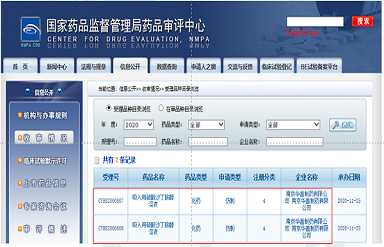
The Salbutamol Sulfate Inhalation Solution (2.5ml:2.5mg ) and the Salbutamol Sulfate Inhalation Solution (2.5ml:5mg ), which researched and developed independently by Aureole and is the first product to register, receives the reference number issued by CDE on November 25th, 2020, and the reference number are CYHS2000807 and CYHS200808 respectively.
The Salbutamol Sulfate Inhalation Solution is used to treat the Asthma and chronic obstructive pulmonary disease (Asthma/COPD) and mainly for the treatment of chronic bronchial spasms and severe acute Asthma attack when the traditional treatment is ineffective. It’s administration method allows the drug goes directly into the lungs and take effect, which could reduce adverse reactions and improve drug efficacy.
The Salbutamol Sulfate Inhalation Solution is the only inhalation solution that is included into the category of anti-asthmatic drugs in the National Basic Drug List, and it was recommended by many guidelines and consensus.

